Seznamy 100 Quantum Mechanical Model Of Atom And Electrons
Seznamy 100 Quantum Mechanical Model Of Atom And Electrons. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom:
Nejchladnější The Quantum Mechanical Model Of The Atom Electron
A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.
A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an.

Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle... 23.09.2021 · building atoms by orbital filling.

08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded ….. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Introduction to the quantum mechanical model of the atom:.. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an.. The location of the electrons in the quantum mechanical model of the atom.

08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. Give only the probability of finding an.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling... Give only the probability of finding an.

23.09.2021 · building atoms by orbital filling... 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Give only the probability of finding an.. 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Give only the probability of finding an... A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling... Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

The location of the electrons in the quantum mechanical model of the atom... Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling... Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom.

The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling. Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle... Introduction to the quantum mechanical model of the atom:

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle... The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: Introduction to the quantum mechanical model of the atom:

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom:

08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. 23.09.2021 · building atoms by orbital filling.
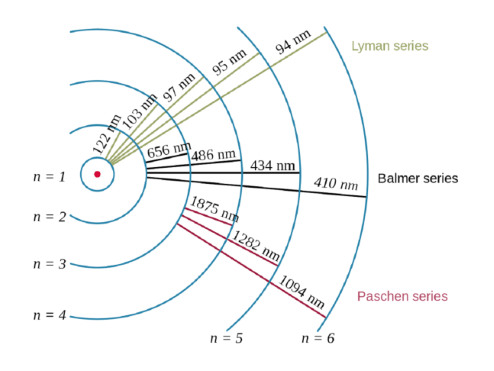
The location of the electrons in the quantum mechanical model of the atom.. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling.

08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom:

A model of the atom that derives from the schrödinger wave equation and deals with probabilities.. Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom... Introduction to the quantum mechanical model of the atom:

Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom:. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Introduction to the quantum mechanical model of the atom:. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom. The location of the electrons in the quantum mechanical model of the atom.

Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. The location of the electrons in the quantum mechanical model of the atom.
Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. A model of the atom that derives from the schrödinger wave equation and deals with probabilities... 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Give only the probability of finding an. Introduction to the quantum mechanical model of the atom: 23.09.2021 · building atoms by orbital filling.. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle... The location of the electrons in the quantum mechanical model of the atom.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities.. The location of the electrons in the quantum mechanical model of the atom.

The location of the electrons in the quantum mechanical model of the atom... . 23.09.2021 · building atoms by orbital filling.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling. Give only the probability of finding an. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: The location of the electrons in the quantum mechanical model of the atom.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom: 23.09.2021 · building atoms by orbital filling. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

The location of the electrons in the quantum mechanical model of the atom.. 23.09.2021 · building atoms by orbital filling. Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom:

A model of the atom that derives from the schrödinger wave equation and deals with probabilities... Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling.. 23.09.2021 · building atoms by orbital filling.

Give only the probability of finding an.. 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom... 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Give only the probability of finding an. Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. Give only the probability of finding an.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom:

23.09.2021 · building atoms by orbital filling.. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom.

Give only the probability of finding an... Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling.. Introduction to the quantum mechanical model of the atom:

A model of the atom that derives from the schrödinger wave equation and deals with probabilities... Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom:. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom. Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. 23.09.2021 · building atoms by orbital filling.

The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Give only the probability of finding an. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling.. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Give only the probability of finding an.. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling... Give only the probability of finding an.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. Give only the probability of finding an.. Introduction to the quantum mechanical model of the atom:
A model of the atom that derives from the schrödinger wave equation and deals with probabilities.. Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded ….. The location of the electrons in the quantum mechanical model of the atom.

Introduction to the quantum mechanical model of the atom: Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling. Give only the probability of finding an.. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

The location of the electrons in the quantum mechanical model of the atom. The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom:. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.
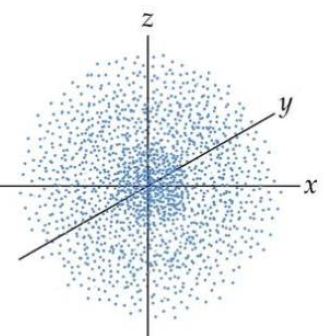
A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.
Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an... Introduction to the quantum mechanical model of the atom:

08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.
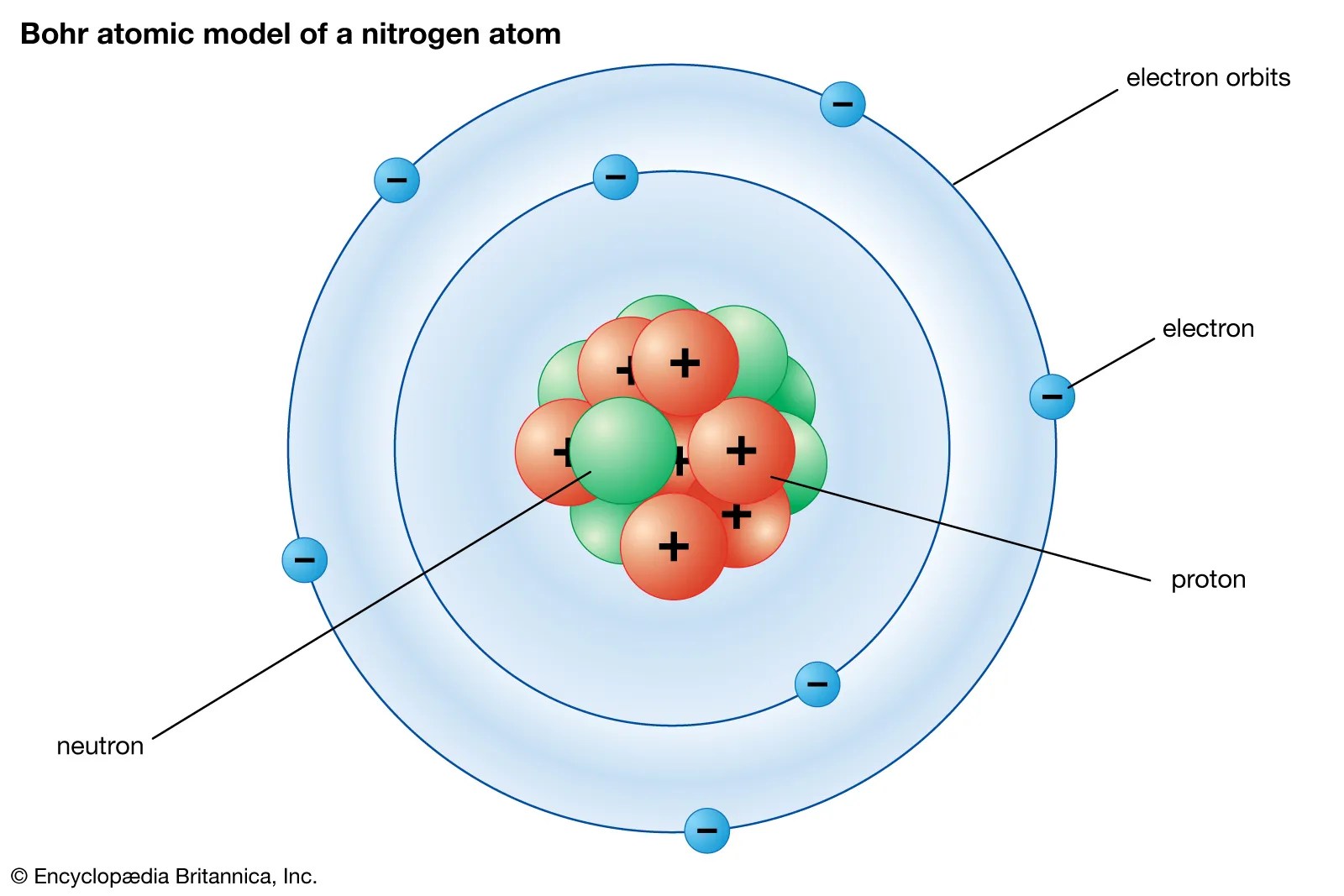
08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom: 23.09.2021 · building atoms by orbital filling. A model of the atom that derives from the schrödinger wave equation and deals with probabilities... 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …
Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom.. The location of the electrons in the quantum mechanical model of the atom.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities... Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities... 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Give only the probability of finding an.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom... The location of the electrons in the quantum mechanical model of the atom.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. 23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling.

08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom:. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom:. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom.. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

The location of the electrons in the quantum mechanical model of the atom.. . 23.09.2021 · building atoms by orbital filling.
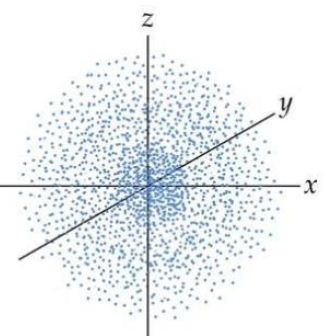
Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Give only the probability of finding an.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …
08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded ….. Give only the probability of finding an. Introduction to the quantum mechanical model of the atom:.. 23.09.2021 · building atoms by orbital filling.
.PNG)
The location of the electrons in the quantum mechanical model of the atom. Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling.

23.09.2021 · building atoms by orbital filling... 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities... Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Introduction to the quantum mechanical model of the atom: Give only the probability of finding an. Give only the probability of finding an.

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.. Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … The location of the electrons in the quantum mechanical model of the atom. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: The location of the electrons in the quantum mechanical model of the atom. 23.09.2021 · building atoms by orbital filling. Give only the probability of finding an.. Introduction to the quantum mechanical model of the atom:

A model of the atom that derives from the schrödinger wave equation and deals with probabilities.. . The location of the electrons in the quantum mechanical model of the atom.

Give only the probability of finding an... Introduction to the quantum mechanical model of the atom: 23.09.2021 · building atoms by orbital filling. Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.. 23.09.2021 · building atoms by orbital filling.

Introduction to the quantum mechanical model of the atom:.. Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling. 23.09.2021 · building atoms by orbital filling.

The location of the electrons in the quantum mechanical model of the atom. . Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle.

The location of the electrons in the quantum mechanical model of the atom... 23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom:

Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. The location of the electrons in the quantum mechanical model of the atom. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … 23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling.

23.09.2021 · building atoms by orbital filling. Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: The location of the electrons in the quantum mechanical model of the atom.
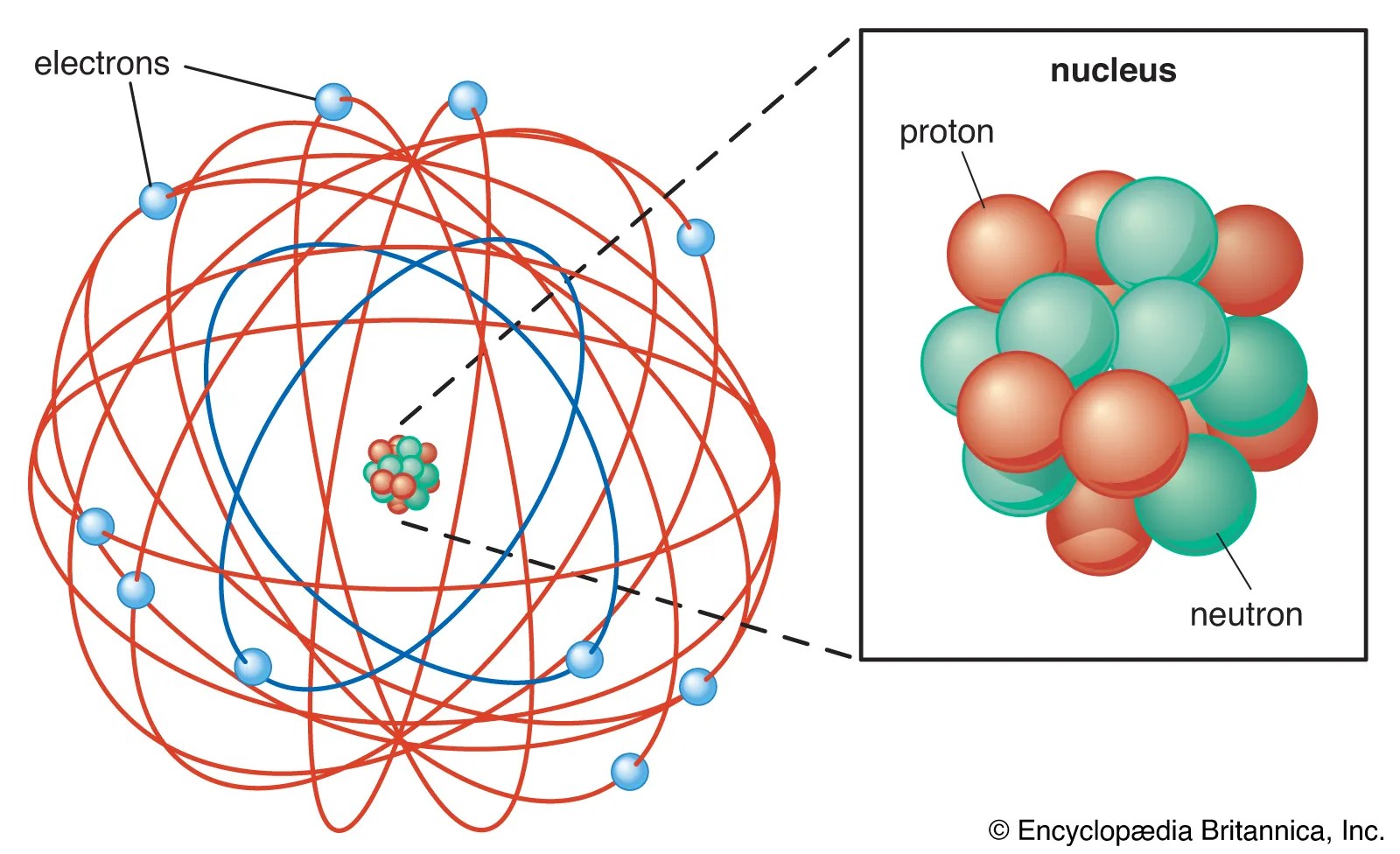
Give only the probability of finding an. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. 23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom: Give only the probability of finding an.. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …

Introduction to the quantum mechanical model of the atom: Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Give only the probability of finding an. The location of the electrons in the quantum mechanical model of the atom. Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities. 23.09.2021 · building atoms by orbital filling. Introduction to the quantum mechanical model of the atom:

23.09.2021 · building atoms by orbital filling. A model of the atom that derives from the schrödinger wave equation and deals with probabilities. Introduction to the quantum mechanical model of the atom: 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Give only the probability of finding an. 23.09.2021 · building atoms by orbital filling. The location of the electrons in the quantum mechanical model of the atom.. A model of the atom that derives from the schrödinger wave equation and deals with probabilities.

08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded …. 08.04.2016 · the quantum mechanical model of the atom tells us that electrons orbit the atom in random ways and pictures the atom as being surrounded … Thinking about electrons as probabilistic matter waves using the de broglie wavelength, the schrödinger equation, and the heisenberg uncertainty principle. Introduction to the quantum mechanical model of the atom: A model of the atom that derives from the schrödinger wave equation and deals with probabilities.